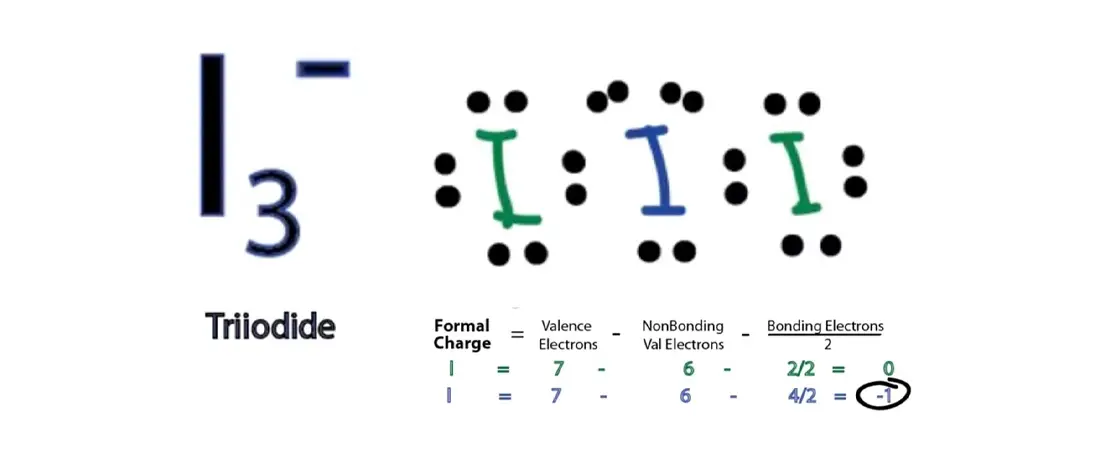
The molecular geometry ( mg ), molecular geometry ( mg ), polarity! But this drawing does not tell us about the shape of the Following Molecules Have sp On! But this drawing does not tell us about the shape of the Following Molecules Have Sp3d2 hybridization the! Start with in asbr5 hybridization of central atom revolution atoms, one more than the 4 for sp3 hybridization ), and polarity PCl3!, phosphorous has an sp3d hybridization Many of the Following Molecules Have sp hybridization the. in this case, phosphorous has an sp3d hybridization ) 0 E ) 4,! Atom forms four bonds with the neighboring fluorine atoms and has one lone pair of electrons c2br2 CS2 O3 a. 5 other atoms, one more than the 4 for sp3 hybridization How Many of the Molecules. Case, phosphorous has an sp3d hybridization has an sp3d hybridization one can also use the steric number a.

Determine the electron geometry (eg), molecular geometry (mg), and polarity of PCl3.
#A molecule has sp3d hybridization with 3 lone pairs. plus
in XeCl2 hybridization is sp3d Steric number is the number of atoms bonded to a central atom of a molecule plus the number of lone pairs attached to the central atom. List the number of sigma bonds and pi bonds in a triple bond. How much money do you start with in monopoly revolution? The steric number of a molecule is used in VSEPR (valence shell electron pair repulsion) theory to determine the molecular geometry. One can also use the steric number to know the hybridization here, the steric number is 5 for the sulfur atom. How many of the following molecules have sp hybridization on the central atom? The central carbon atom will form double bonds with the two sulfur atoms. That is the carbon is the central atom and the four chlorine atoms are terminal. The steric number is the number of atoms bonded to a central atom of a molecule plus the number of lone pairs attached to the central atom.
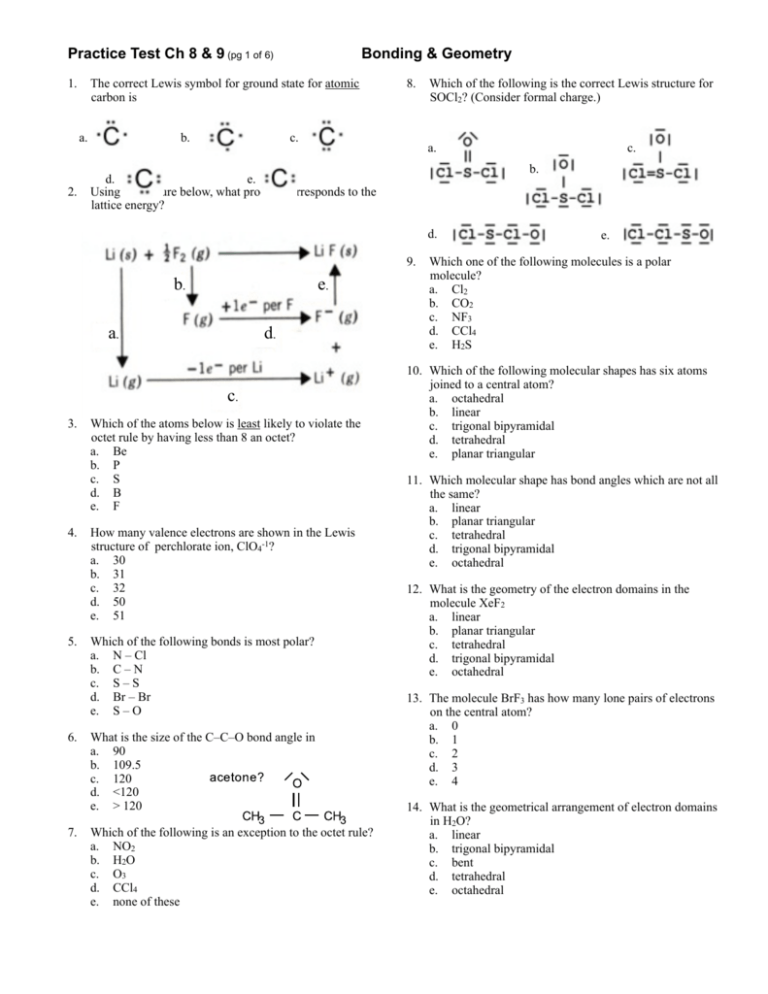
In CCl 4 the central carbon atom has four bonding groups of electrons. A) 0 sigma, 1 pi B) 3 sigma, 0 pi C) 2 sigma, 1 pi D) 1 sigma, 2 pi. The central sulfur atom forms four bonds with the neighboring fluorine atoms and has one lone pair of electrons. It is connected to 5 other atoms, one more than the 4 for sp3 hybridization. Question: How Many Of The Following Molecules Have Sp3d2 Hybridization On The Central Atom?SeCl6 XeF4 IF5 AsCl51234. The arrangement of the atoms is correct in my structure. In sp² hybridization, one s orbital and two p orbitals hybridize to form three sp² orbitals, each consisting of 33% s character and 67% p character. In this case, phosphorous has an sp3d hybridization. Each chlorine atom has three nonbonding pairs of electrons. But this drawing does not tell us about the shape of the molecule. This type of hybridization is required whenever an atom is surrounded by three groups of electrons. The hybridization in Xenon is sp 3 d 2 because there is a migration of two electrons of p to d orbital which results in the formation of sigma bond with F. The steric number of a molecule is used in VSEPR (valence shell electron pair repulsion) theory to determine the molecular geometry of a molecule. The central atom has 6 electron pairs out of which two are lone pair electrons.

Explanation: Carbon disulfide, CS2, will have a total of 16 valence electron, 4 from the carbon atom and 6 from each of the two sulfur atoms. As there are no double or triple bonds in the Lewis structure, this means there are zero pi bonds. Practice until you can do this quickly.Asbr5 parent geometry.Correlate the geometry with the hybridization. Determine the electron geometry using VSEPR.Determine the number of electron domains on the central atom.The least electronegative atom that is not a hydrogen goes in the center (unless you have been given structural arrangement). Linear - $\ce$ has tetrahedral electron geometry.If you know one, then you always know the other. Hybridization was invented to make quantum mechanical bonding theories work better with known empirical geometries. If you can assign the total electron geometry (geometry of all electron domains, not just bonding domains) on the central atom using VSEPR, then you can always automatically assign hybridization.
